Download PDF of Technical Paper: Novel Technology of Electron Beam Curing in Vacuum
Mikhail Laksin, IdeOn LLC, Bowei Han Celplast Metallized Products and Dante Ferrari, Metallizing Surface Technologies
Abstract
The idea of applying and curing functional coatings using Electron Beam in vacuum has been explored for many years with limited commercial success. Significant practical benefits of applying very thin (less that one micron thick) EB curable coatings directly on vacuum-metalized plastics have been seriously undermined by difficulties with coating application technique, which employs spraying of low viscosity radiation curable liquid blends. Such blends usually have limited stability in vacuum. These problems have been resolved by introducing Flexographic printing/coating method inside of the vacuum metallization chamber and optimizing coating chemistry for enhanced stability in vacuum. The new process allows application of equally low film thickness without traditionally excessive loss and polymerization of the sprayed coating inside of the vacuum chamber. This new process also offers enhanced degree and speed of crosslinking by taking advantage of Plasma and Ultra Violet energy, in addition to vacuum generated EB irradiation. Such films demonstrate a range of improved performance attributes; the protective coating enhances moisture and gas barrier properties, reduces corrosion, increases lamination bonds and offers low migration, under threshold of FDA regulations. This paper discusses benefits of the new technology that make application of these coatings a viable option desirable in a broad range of market segments utilizing vacuum metalized plastics.
Introduction
Electron Beam (EB) curing is a powerful tool, used to form thin protective coatings for various organic and inorganic surfaces, Including wood, polymer films and metals. The most efficient application methods for thin, 2-3 micron or less, coatings are Flexographic (Flexo) and Gravure printing. These methods offer high manufacturing throughput as the application can take place at very high speeds up to 200-400 m/min. In most cases, Flexo or Gravure applied coatings satisfy various decorative requirements such as gloss that can be high or low, while offering enhanced abrasion and chemical resistance to the coated material. The major challenge is to apply uniform and defect free coating layers over a broad range of materials, varying in surface energy and micro-roughness/topography.
The fundamental limitation of both Flexo and Gravure printing is that the coating metering is based on transferring of liquid coating from individual cells of Anilox or Gravure cylinder. It is expected that once small portions/droplets of the coating are transferred from the cells onto the substrate, the liquid would quickly spread out, forming a continuous layer prior to EB curing. Such spreading is driven by the forces of gravity and viscous flow, working against surface energy forces that prevent liquid for spreading. Another words, liquid spreading is controlled by the gradient between surface energy of the substrate and surface tension of the liquid coating. If surface energy of the substrate is too low and surface tension of the coating is too high, spreading is limited, leaving significant defects such as voids and pinholes in the finished coating. These defects diminish protective properties of the coating especially when high chemical or moisture resistance are expected. It is relatively easy for aggressive chemicals or moisture to penetrate such porous coating and attack the substrate.
Protection of metallized surfaces such as aluminum foil or vacuum metalized plastics is especially challenging. Aluminum Oxide on the surface of these materials is difficult to adhere to. The combination of voids and limited adhesion to the hard and chemically inert surface significantly limits the mechanical and chemical protection expected from EB coatings.
It has been suggested to apply EB coatings directly in the vacuum chamber by spraying liquid coating into the vacuum over the substrate and cure it with an EB source. Vapor deposition process involves the evaporation of a liquid blend in a vacuum chamber, its deposition onto a cold substrate, and subsequent polymerization by exposure to electron beam. Typically, a liquid coating from a supply reservoir delivered to a heated evaporator section of a vacuum deposition chamber where it flash vaporizes under vacuum1,2,3. The resulting reactive vapor passes into a condensation section of the unit where it is vapor applied onto a substrate, condenses and forms a thin liquid film upon contact with the cold surface of the substrate. The liquid deposited film is then cured by exposure to an electron beam. In this case, EB curing requires significantly lower accelerating voltage than is used in a typical E-beam to cure coating outside vacuum. Applying coating inside vacuum metallization chamber over the not yet oxidized aluminum surface offers greater uniformity and enhanced adhesion. Unfortunately, spraying of coating into the vacuum at 104 to 106 Torr, typical for vacuum metallization process, causes significant losses of coating that is spreading over entire inner surface of the vacuum chamber, often getting inside of the vacuum pump. The coating that ends up on the walls of the vacuum chamber gets gelled/polymerized and requires significant removal efforts after each metallization and coating/curing cycle.
Alternative Process of EB Curing in Vacuum
It was suggested to introduce Flexographic process of applying a liquid coating inside of the vacuum chamber 4. This approach eliminated loss of the coating completely. The transfer of the coating takes place in vacuum. As a result, spreading of the liquid over freshly metalized substrate is more complete and the coating has higher uniformity and lower number of defects. In addition, surface energy of fresh Al layer is very high which, in turn, creates a significant surface energy gradient, beneficial for effective spreading of coating.
This process, as shown schematically in Figure 1, involves:
- unwinding the film under vacuum, metallizing the film surface,
- in-line coating using a flexographic process,
- curing with E-beams,
- rewinding the film under vacuum,
- venting the chamber to atmosphere
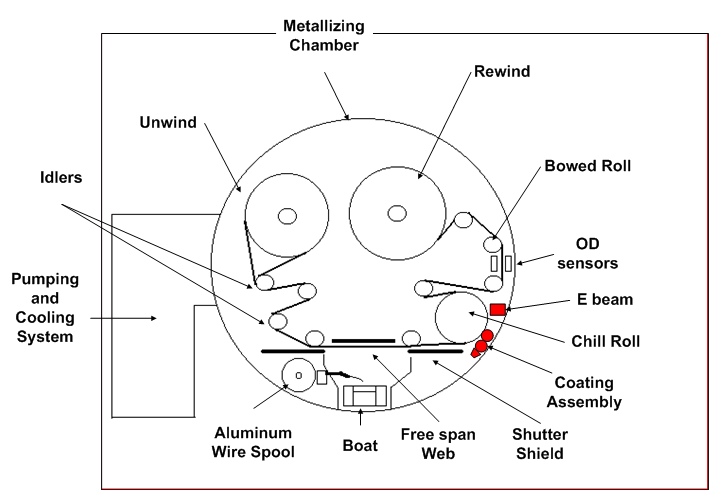
Figure 1 Coating and EB curing in Vacuum Metallization Chamber
In was demonstrated in the work by J, Weiser and others5, that electron beam at 20 keV used for the excitation of pure rare gases such as argon, krypton, and xenon at pressures up to 1.7bar can provide an efficient light source in the vacuum ultraviolet spectral region between 120 and 200 nm. In this work, Electron beam, delivered through a thin foil, into a purified gas filled chamber, was used only as an excitation tool to generate UV light.
Curing of coatings induced by Plasma is suggested by Misev et al.6. In this case, a 3-dimensional object is placed in a Plasma discharge chamber and initial curing takes place upon exposure to plasma treatment. Additional post-thermal treatment is recommended to complete curing.
It is suggested that as voltage is applied to a Tungsten electrode inside of the vacuum chamber in presence of gas flow, directing electrons towards the Flexo applied coating, it simultaneously generates three energy sources, inducing polymerization – Electron Beam, Plasma and UV light. It was found that all three energy sources have a positive synergistic effect on degree of polymerization and cure speed of the coating7.
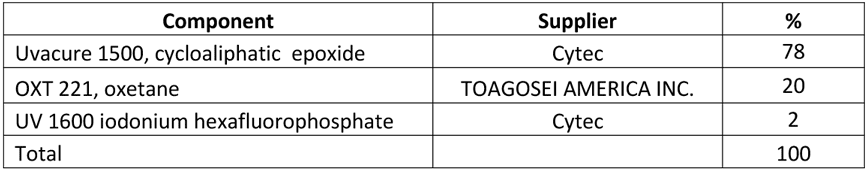
Selection of a gas, directing flow of electrons in vacuum appears to be an important factor in generating sufficiently strong Plasma and UV radiation, accelerating EB curing. This can be illustrated by the following examples. It is known that cationically curable compositions, such as those presented in Table 1, can undergo EB polymerization in presence of iodonium salt based photoinitators.
Table 1 Cationic composition for curing in vacuum
This composition was applied over vacuum metalized polyester film in presence of different gases. Degree of cure was assessed by amount of back transfer of coating in the roll of coated film at different line speeds. The results are summarized in Figure 2.
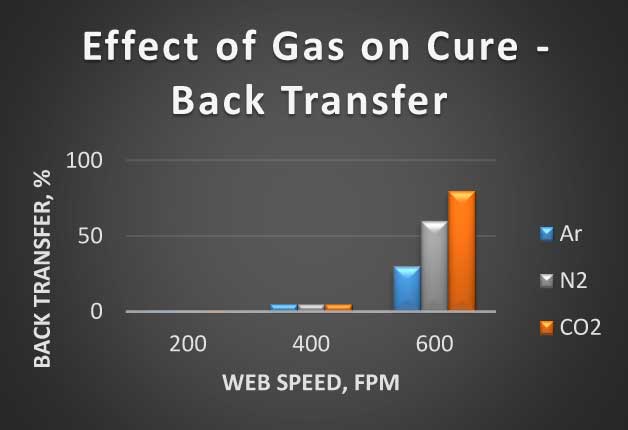
Figure 2 Effect of gas selection on curing
Based on the back transfer evaluation, Argon is the most effective gas, following by Nitrogen. The largest back transfer took place at 600 fpm of web speed with CO2.
Effect of gas selection on chemical resistance, as measured by double IPA (Isopropanol) rubs required to visually impact the coating, is illustrated by Figure 3.
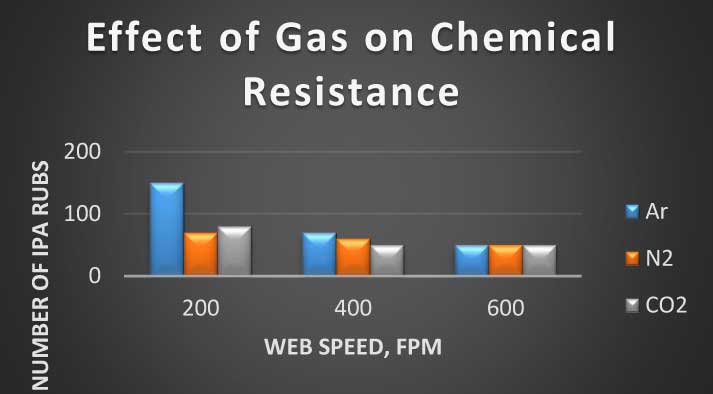
Figure 3 Effect of gas selection on crosslinking
Argon demonstrates significantly higher chemical resistance at 200 fpm of web speed but it appears that the difference between the gases is diminishing with web speed and at 600 fpm chemical resistance for all gasses is about the same at 50 IPA Rubs.
In order to confirm participation of Plasma and UV irradiation in curing, a composition that is not capable of EB curing in air under normal atmospheric pressure was used in the vacuum process. The composition is presented in Table 2.
Table 2 Cationic composition for curing in vacuum
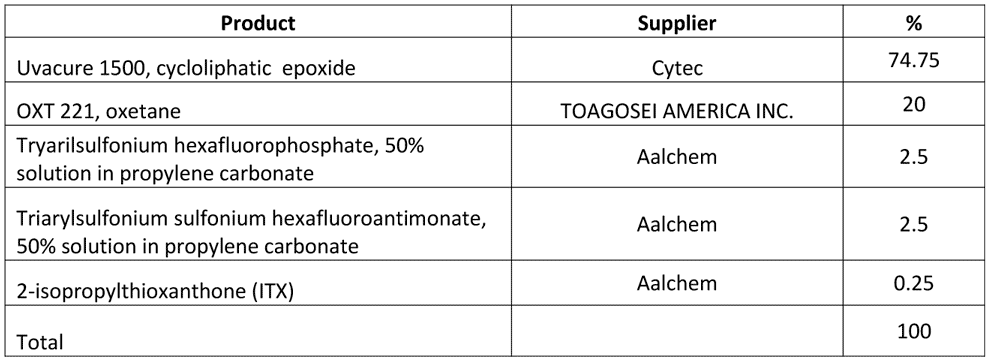
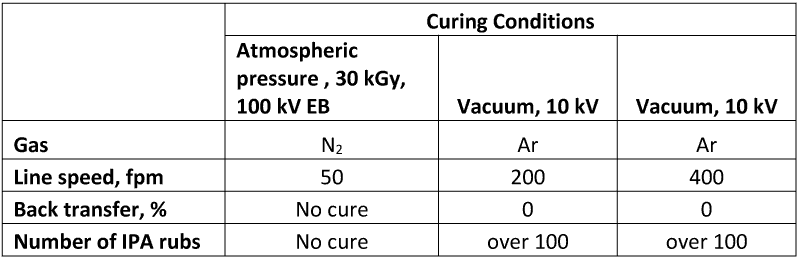
This composition remains liquid when exposed to 30 kGy of EB dose at 100 kV in AEB laboratory curing unit. When tested in vacuum in presence of Argon, this composition was completely cured at relatively high web speed without back transfer and with very high chemical resistance. The curing results are summarized in Table 3.
Table 3 Comparison of cure under atmospheric pressure and vacuum
Post-Cure Migration Study
Metalized films are commonly used in Food Packaging as a moisture and oxygen barrier. Introduction of EB curable protective coating over Aluminum layer is considered to be one of the key applications for this technology. It is important to assess degree of crosslinking and potential migration of unreacted compounds on both sides of the coated film by conducting extraction and analytical characterization of the extract. This work was conducted at Rutgers University in Center for Advanced Food technology by Dr. Thomas Hartman.
Polyester Film was metalized and coated with EB coating in vacuum chamber at …… m/min web speed. The analysis of both for coated and uncoated sides was done according to FDA extractables testing guidelines using 95% ETOH and FDA Condition of Use E extraction parameters (10 days at 40°C).
Sections of sample were cut and placed into custom stainless steel (SS) extraction cells designed according to FDA specifications for food contact migration testing. The cell is composed of two SS plates which sandwich a Teflon gasket assembly. The gasket isolates only the food-contact side for extraction and contains a cavity to hold the extracting solvent. The surface area exposed for extraction within the cavity was 51 cm2 (7.9 in2 and 79 ml of 95% ETOH was used as the extraction solvent. Therefore, the solvent volume surface area ratio was 1.55 ml/cm2 or 10 ml/in2. The samples were extracted as per FDA Condition of Use E (10 days at 40°C) from both the coated or uncoated side. Following extraction the ETOH extracts were matrix-spiked with 1000 ppb w/v (79 μg) of anthracene -d10 to serve as an internal standard and back-extracted into methylene chloride. The extracts were then concentrated to approximately 1.0 ml using a gentle stream of nitrogen at room temperature. The concentrated extracts were then analyzed by GC-MS. The resulting GC-MS data from all samples were subjected to a thorough scan-by-scan examination and all compounds in the extracts were identified and/or characterized.
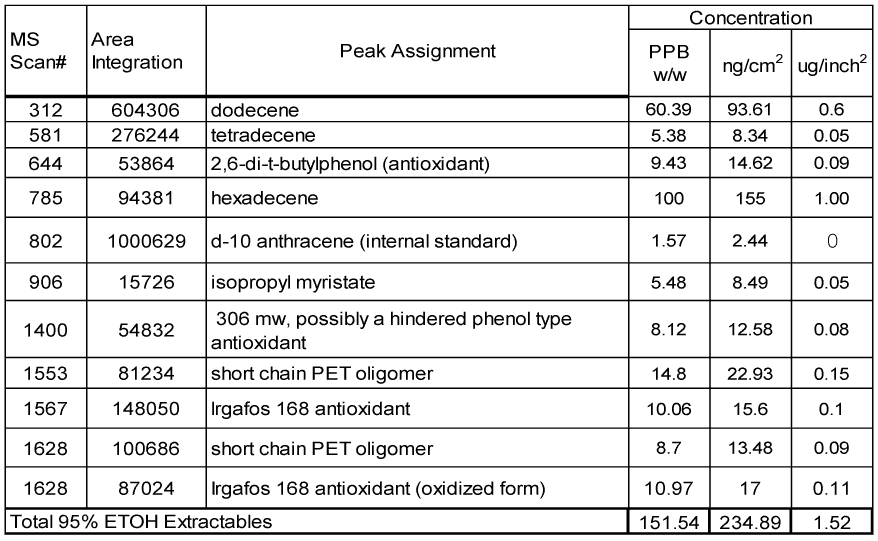
The data for the extractables of the samples are summarized in Tables 4 and 5. The Tables provide complete peak assignments and concentration data for extractables. From left to right, each Table lists the MS scan number (from centroid of peak), peak area integration, peak identification and then concentration data expressed in three different units (ppb w/v, ng/c1n2 and μg/inch2). The first column of concentration data is the migrants in parts per billion on a weight to volume basis (ppb w/v) in the extraction solvent. The second column of concentration data is expressed in nanograms of migrant per square centimeter (ng/cm2) of surface area exposed to extraction and the final column of concentration data is expressed in micrograms of migrant per square inch (μg/inch2)
The sample tested extremely clean from both the coated and uncoated side. No EB-cure chemistry related extractables were detected at all. Table 4 illustrates total migration from uncoated metalized PET film. Small quantities of various compounds were detected, most of them with exception of long chain dodecene were under 50 ppb.
The same compounds at about the same levels were detected in the extract from the EB coated metallized PET film – Table 5. No detectable amount of any EB coating component is detected in this test.
Table 4 – Uncoated Metallized Film Extraction
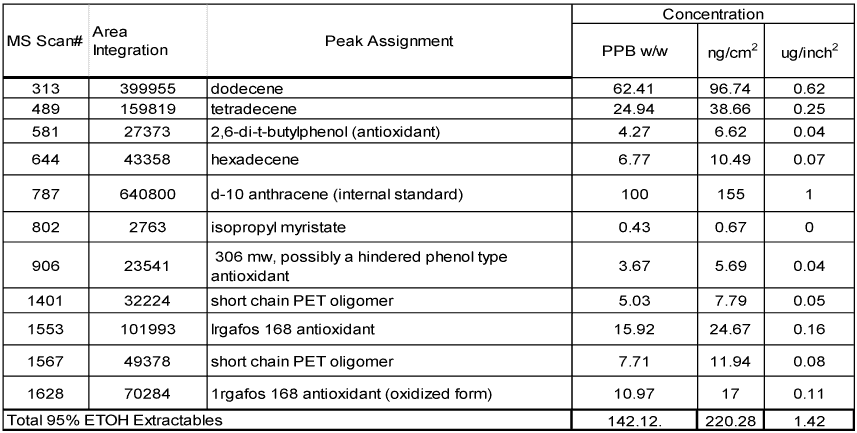
Table 5 – Coated side Extraction
Similar results have been obtained in multiple migration studies suggesting that curing in vacuum allows for a high degree of conversion of reactive components of the coating.
Application to metallized films
Barrier Improvements
EB coating under vacuum has been applied on a small commercial scale by Celplast Metallized Products. The results have shown that applying in-line in-vacuum EB curable coating immediately after the metallization zone can enhance the barrier properties of metallized film by up to an order of magnitude.
In typical metallized film production, according to Pinhole Theory the gas transmission is governed by the number of defects in the metal layer. This is schematically shown in Figure 4, with the yellow layer being the film layer and the blue layer being the vapor deposited aluminum layer.
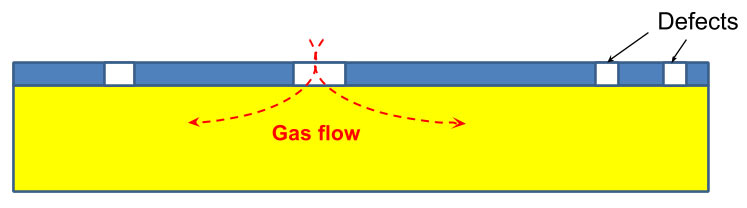
Figure 4: Film Defects
These defects are usually caused by scratches and impurities in the winding process. Debris and impurities create areas of low metal adhesion that can lead to pinholes, as shown in Figure 5.
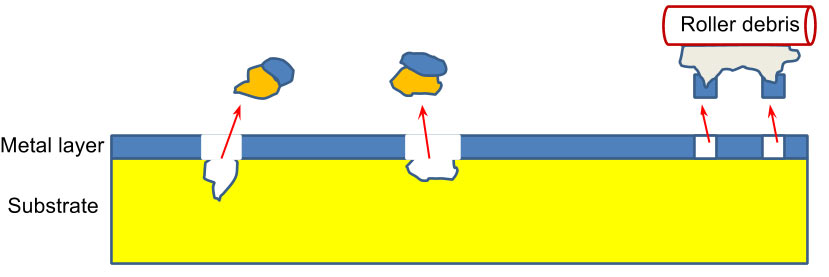
Figure 5: Metal pick-off
The traditional two-pass method for preparing top-coated metallized films using roll-to-roll processes involves:
- Unwinding the film under vacuum, metallizing the film surface, rewinding the film under vacuum, venting the chamber to atmosphere.
- Unwinding the film in atmosphere, top coating the metallized surface, rewinding the film in atmosphere.
This process applies a top coat over metallized films where the pinhole defects have already occurred. In contrast, the in-line metallizing and top-coating process can be conducted sequentially in a single pass.
This process prevents pinholes because any surface impurities that can flake off will be trapped immediately by the coating after metallizing. This makes it possible produce higher quality films with few defects, as seen in Figure 7.

Figure 6: Back-lit metallized films with & without in-line top-coating
Several base film types have been metallized and coated using this process at various optical densities. Barrier measurements were taken to compare the performance of metallized films that were in-line coated vs. uncoated. The results are shown in Table 6 (WVTR) and Table 7 (OTR). Note that in each case the metallized film barrier was compared to the top-coated metallized film in runs where the coating nip was engaged, then dis-engaged, so the base film type and metallization conditions were identical in each case.
Table 6: Improvements in WVTR with in-line top-coating technology*
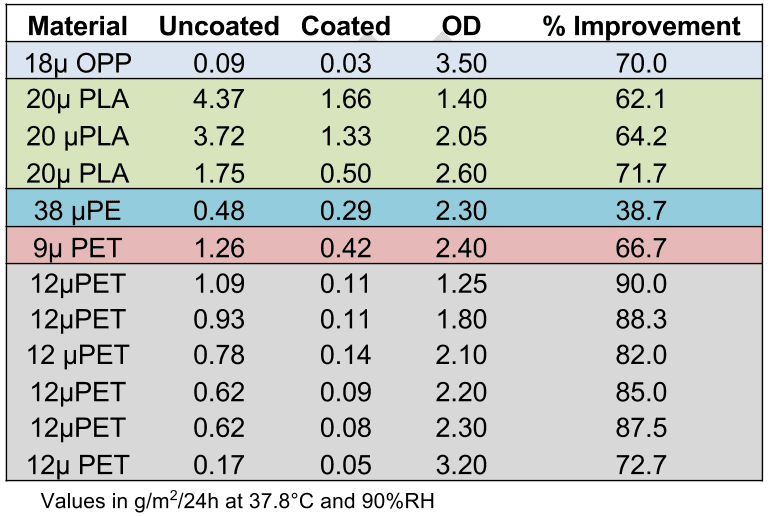
Table 7: Improvements in OTR with in-line top-coating technology*
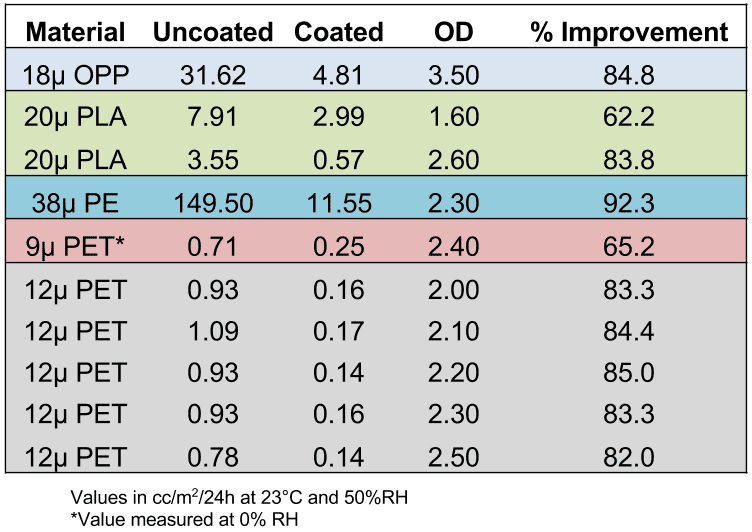
*Standards for barrier properties of metallized films are based on WVTR (water vapor transmission rate) and OTR (oxygen transmission rate). WVTR is measured in accordance with ASTM E-398 at 37.8C and 90% relative humidity. OTR is measured in accordance with ASTM D-3985 at 23C and 50% relative humidity.
The results show that it is possible to significantly improve barrier properties by metallizing and EB coating in a single step, even at lower optical densities. This is regardless of base film type.
Sensitivity to Downstream Processing
Consumer goods companies are becoming more aware of the fact that barrier properties degrade as packages travel through the supply chain due to handling during storage, transportation and usage. An additional advantage of EB coated metallized films is their resistance to downstream processing. Gelbo flex tests were performed to compare the barrier degradation (OTR) of EB coated metallized films versus uncoated metallized films in a common 3-ply laminate structure (printed PET/met PET/sealant web). Three different adhesive lamination techniques were tested and in each case the EB coated material was shown to be less susceptible to barrier degradation (Figure 8).

Figure 8: Oxygen barrier properties of 3-ply laminates with HB Met PET vs. Coated Met PET, before and after Gelbo flexing, 3 different lamination techniques
Adhesion and lamination bond strengths
Metal to film and coating to metal bond strengths must be suitable. The industry accepted method for bond strength is to use Scotch 610 or 810 tape, pulled back at a 180° angle from the film surface. A pick- off level of <1% is suitable for most applications. This bond level is difficult to achieve with atmospheric EB coating – perhaps because the oxidized aluminum surface, although having a high surface energy, requires aggressive acidic or basic chemistries to achieve good bonding with the coating. However, under vacuum with minimal oxidation of the aluminum surface, it was found that <1% pick-off can be achieved with a variety of EB chemistries.
Corrosion resistance
Corrosion resistance is essential for markets such as building insulation. This is measured by directly exposing the coated metallized surface to elevated heat and humidity (71C, 100%RH) for seven consecutive days and then carrying out a subjective visual evaluation of the level of corrosion of the metal surface, in accordance with ASTM D3310. We have found that it is possible to achieve suitable corrosion resistance with certain EB chemistries as long as the coating is applied consistently.

Figure 9: Two test specimens on light box with 0.2 gsm coating after D3310 testing
Conclusions
Introduction of EB curing inside of the vacuum metallization chamber offers significant benefits in improvement of barrier, mechanical and chemical (corrosion) resistance properties of metalized products. These improvements are achieved with relatively thin films due to a high degree of uniformity of the coatings applied in vacuum and excellent adhesion to the aluminum surface that has not been oxidized. The curing takes place in the presence of several energy sources including EB, Plasma and UV, offering a possibility of using a range of options for selecting coating chemistry from free radical to cationic systems. A high degree of curing conversion allows one to produce coatings with practically undetectable extractables, making this process very attractive for various food packaging applications.
References
1. Michael Mikhael and Angelo Yializis “Thin coating manufactured by vapor deposition of solid oligomers”, U.S. Patent 6,270,841, July 2, 1999
2. Michael Mikhael and Angelo Yializis “- Vacuum-deposited colorants “, US Patent 6,447,553, September 10, 2002
3. Michael Mikhael and Angelo Yializis “Vacuum deposition of cationic polymer systems” US Patent 6,468,595 – August 3, 2005
4. Mikhail Laksin, Wolfgang Decker and Subhankar Chatterjee “Electron beam curable composition for curing in vacuum chamber” US Patent 8,460,762 , June 11, 2013
5. J. Wieser and others – Vacuum ultraviolet rare gas excimer light source Rev. Sci. Insrum., 68(3), March 1997
6. Ljubomir Misev, Anreas Valet, Peter Simmenbinger and Tunja Jung “Curing of coating induced by plasma” US Patent 8,003,169, August 23, 2011
7. Mikhail Laksin, Wolfgang Decker and Siddharth Fernandes “Process for Curing a Composition by Electron Beam Radiation, and by Gas-Generated Plasma and Ultraviolet Irradiation” US Patent 9011983, April 21, 2015
Download PDF of Technical Paper: Novel Technology of Electron Beam Curing in Vacuum

